

Next: Experiments and results
Up: Applications of recurrence quantified
Previous: Introduction
The peroxidas-oxidase (PO) reaction is an important bridge between the chemical
excitable oscillators Beluzov Zabotinsky reaction and biological oscillators
such as intracellular
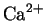 oscillators [6].
It is now clear that PO reactions shows a wide spectrum of interesting
behaviors including simple oscillations, bistability, quasiperiodicity and
chaos [7].
These behaviors have all been observed in vitro under well controlled
laboratory conditions. This reaction appears in plants as part of the process of
lignifications [8] with nicotinamide adenine dinucleotide (NADH) as electron
donor. Its estoqueometric formulae is:
oscillators [6].
It is now clear that PO reactions shows a wide spectrum of interesting
behaviors including simple oscillations, bistability, quasiperiodicity and
chaos [7].
These behaviors have all been observed in vitro under well controlled
laboratory conditions. This reaction appears in plants as part of the process of
lignifications [8] with nicotinamide adenine dinucleotide (NADH) as electron
donor. Its estoqueometric formulae is:
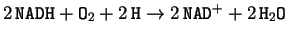 .
In 1983, a model of PO reaction, now commonly referred as
the Olsen's Model [9], was proposed. Simulations with the Olsen model
quantitatively reproduce both
the simple and chaotic oscillations of PO reactions. Studies with this model
showed that increasing the parameter
.
In 1983, a model of PO reaction, now commonly referred as
the Olsen's Model [9], was proposed. Simulations with the Olsen model
quantitatively reproduce both
the simple and chaotic oscillations of PO reactions. Studies with this model
showed that increasing the parameter 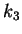 caused the system to undergo
a transition from simple oscillations to chaos via a cascade of periodic
doubling bifurcations. The Olsen's model involves four variables,
molecular oxygen A,
NADH B and two intermediate species. One of the intermediates is very likely
caused the system to undergo
a transition from simple oscillations to chaos via a cascade of periodic
doubling bifurcations. The Olsen's model involves four variables,
molecular oxygen A,
NADH B and two intermediate species. One of the intermediates is very likely
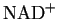 X, while another one is oxyferrous peroxidase Y,
also known as compound III.
The complete mechanism corresponds system of four differential equations
are given below, with
X, while another one is oxyferrous peroxidase Y,
also known as compound III.
The complete mechanism corresponds system of four differential equations
are given below, with 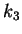 as control parameter which induces a transition to chaos
type I [10].
as control parameter which induces a transition to chaos
type I [10].


Next: Experiments and results
Up: Applications of recurrence quantified
Previous: Introduction
Horacio Castellini
2004-10-27